Welcome back Vitalians! We’re excited to share our monthly update on everything happening across the VitaDAO ecosystem and the wider longevity community.
But first, please let us take a moment to reflect upon the passing of Craig Venter.

The sequencing of the human genome is one of humanity's landmark achievements. Craig Venter accelerated the Human Genome Project by pursuing a radically different strategy: instead of the publicly funded consortium’s slower, map-based sequencing, he used whole-genome shotgun sequencing at scale through Celera Genomics, combining high-throughput sequencing with aggressive computational assembly to produce a draft human genome years ahead of schedule.
Beyond that race, he was a central figure in early gene discovery (notably via expressed sequence tags), later founding the J. Craig Venter Institute, led the first creation of a cell controlled by a chemically synthesised genome (JCVI-syn1.0), and helped launch the field of synthetic biology. He also drove large-scale environmental genomics, sampling ocean microbes aboard his vessel Sorcerer II, vastly expanding the known diversity of life and its genetic repertoire. Craig Venter was a titan of modern science whose legacy will live on.
In more uplifting news, our very own VitaRNA CSO, Michael Torres, has just sold CrossBridge Bio (a company he co-founded) to pharma giant Eli Lilly in a deal worth up to $300 million. He is now shifting more of his focus to VitaRNA and its spin-out company, ARTAN Bio, to develop next-generation codon suppressor therapies for rare diseases and aging.
VitaDAO Portfolio Highlights
To date we have funded more than 30 projects. We are always looking out for more ground-breaking longevity drug discovery science to support, so email us at discovery@vitadao.com if you have, or know any researchers with, a great translational idea in need of funding!
Rapamycin & Exercise — Brad Stanfield
This project is a double-blind, placebo-controlled study of weekly rapamycin combined with exercise in older adults, led by Dr. Stanfield with Dr. Matt Kaeberlein as senior scientific collaborator, and co-funded by VitaDAO. This trial, RAPA-EX-01, took place last year, but the results were under peer review. They were finally published on April 15, 2026 in the Journal of Cachexia, Sarcopenia and Muscle.
The 13-week study enrolled 40 sedentary adults aged 65 to 85 who were randomized one-to-one to weekly oral sirolimus 6 mg or matched placebo, alongside a standardized home-based resistance and endurance exercise programme performed three times per week.
The primary endpoint was the change in 30-second chair-stand repetitions at week 13, analyzed by intention-to-treat ANCOVA with adjustment for baseline performance, age stratum, and sex. Both groups improved their performance, but the rapamycin arm completed approximately two fewer repetitions than placebo at the end of the trial, and sensitivity analyses suggested a modest attenuation of functional gains rather than the expected enhancement.
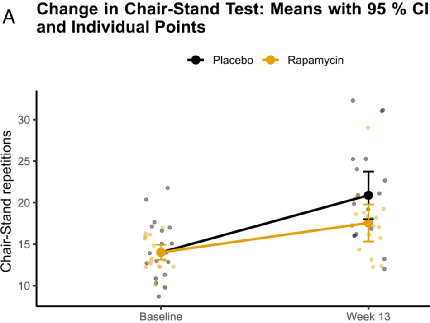
Secondary functional outcomes including 6-minute walk distance and grip strength also numerically favored placebo without reaching statistical significance, and SF-36 quality-of-life scores showed similarly small non-significant differences. Exploratory analyses of C-reactive protein and several epigenetic age measures were inconclusive, and the CRP signal was driven by two outliers in the treatment arm.
Adverse event burden was higher with sirolimus than with placebo (99 events versus 63 across 17 participants in each arm), including one possibly drug-related serious adverse event of pneumonia.
The authors interpret the data through the lens of the cycling hypothesis, which proposed that spacing weekly rapamycin dosing far enough from training sessions would preserve the muscle adaptation response. With a half-life of about 62 hours, a 6 mg dose taken on day 6 leaves substantial drug on board during a day-1 workout the following week, and Stanfield acknowledged that the dosing schedule may not have created a sufficiently clean off-period for the hypothesis to be properly tested. The paper concludes that future trials with longer treatment duration or less frequent and lower dosing are needed to determine whether a favourable benefit-risk profile can be achieved with rapamycin in this setting.
This is the first properly designed, double-blind, placebo-controlled randomized trial of rapamycin combined with exercise in older adults, and we are pleased to have helped catalyze its delivery. The result is an important reality check for the longevity community, where rapamycin has often been discussed on the basis of animal data and self-experimentation rather than rigorous human evidence. A negative or attenuating signal from a single small trial does not close the question, but it does usefully constrain the dosing-and-timing regimens that warrant further investigation. We expect to see follow-on trials testing alternative schedules and lower doses, and the open data from RAPA-EX-01 should inform their design.
VitaLabs — our in-house Longevity Venture Studio

Season 1 of VitaLabs tested a new idea: give creative scientists the means and the freedom to pursue their own longevity research, let a community decide which projects to back, and see if the most important work would find its way through.
Two fellowships came out of that first season. Synthetic Vitality looked at drug-combination effects on lifespan. HSC Rejuvenation mapped the transcriptional landscape of blood-stem-cell aging. Both projects ran cleanly. Looking back, the work sat in a middle ground: too applied to be pure discovery, too exploratory to anchor a company. We've come away with a sharper standard. Every project we fund should be tethered to a real problem we care about (importance), and every result—positive or negative—should narrow the path to a solution (decisiveness).
When we designed season 2, we put more of our attention on decisiveness. We reimagined the application process by pairing a deep, internal review with an interactive revision loop for applicants. It worked. Each proposal became a better version of itself.
But no amount of revision could make the underlying problem more important.
So we’re focused on a different question now: what does VitaLabs look like when the most important questions in longevity come first, and the proposals come after? Stay tuned.
News and Media
We will now shift gears to highlight what has been happening in the wider longevity ecosystem.
The Impetus Longevity grants are revving up for Round 4, with a focus on funding the next generation of shared datasets to power biological AI.
You will have heard debates around whether research should be geared towards lifespan or healthspan, now Alex Zhavoronkov introduces “peakspan” as the new metric we should be aiming to extend!
One of the scientific avenues being explored that could extend peakspan is epigenetic reprogramming - here we have an interesting article discussing David Sinclair’s upcoming clinical trial on epigenetic reprogramming in humans.
On this theme, an article from the Harvard Gazette “Rethinking what it means to age as humans live longer and healthier.”
If you’re looking for an interesting podcast to watch, then check out The Optispan Podcast with Matt Kaeberlein: Neuroscientist Reveals What's Actually Working for Brain Longevity (with Dr. Tommy Wood).
Longevity x Pharma
Longevity is receiving more and more interest from pharma lately. But how did this unfold? To answer this, our very own Paolo Binetti put together this brief history of pharma and longevity. What's more, VitaDAO was involved in 3 of these big events either directly or through our portfolio companies.
🕒 The Foundations (2008–2016)
April 2008: GSK announced the acquisition of Sirtris Pharmaceuticals for $720M, a bet on David Sinclair's sirtuin research.
September 2014: AbbVie and Calico Life Sciences (Alphabet Inc.) launched a collaboration worth up to $1.5B; AbbVie's investment reached ~$1.75B by 2022. AbbVie exited in November 2025.
April 2016: AstraZeneca signed a 10-year genomics collaboration with Craig Venter's Human Longevity, Inc., covering up to 500,000 clinical trial DNA samples.
🚀 The Acceleration (2022–2025)
August 2022: Pfizer Ventures joined VitaDAO (VDP-54.1) with a $500,000 contribution in exchange for VITA tokens.
July 2023: Eli Lilly and Company acquired Versanis Bio for up to $1.92B, gaining bimagrumab for obesity and body-composition biology.
December 2023: Eli Lilly partnered with Fauna Bio to identify obesity targets from comparative genomics across hibernating and extreme-adaptation mammals.
July 2024: AbbVie Ventures led Oisin Biotechnologies' $15M Series A first close (VitaDAO portfolio company).
October 2024: Eli Lilly joined a $43M Series A for Arda Therapeutics, advancing targeted cell-depletion therapies for chronic disease.
December 2024: Novartis and BioAge Labs signed a $550M collaboration on aging and exercise biology.
July 2025: Chugai Pharmaceutical Co., Ltd. (Roche group) signed a research and license collaboration with Gero, milestones up to $250M plus royalties (VitaDAO portfolio company).
🌊 The 2025 "FGF21 Moment"
May 2025: GSK announced the acquisition of efimosfermin from Boston Pharmaceutical Industries for $1.2B upfront, $2B total.
September 2025: Roche agreed to acquire 89bio for $2.4B upfront, $3.5B total.
October 2025: Novo Nordisk agreed to acquire Akero Therapeutics for $4.7B upfront, $5.2B total.
🤖 Lilly Leadership (2025–2026)
June 2025: Eli Lilly partnered with Juvena Therapeutics in a deal worth >$650M, targeting muscle health and body composition through a stem-cell-derived secreted protein platform.
June 2025: Eli Lilly announced the acquisition of Verve Therapeutics for up to $1.3B, gaining in vivo gene editing for cardiovascular disease including PCSK9 program VERVE-102.
September 2025: Eli Lilly entered a gene therapy collaboration with Remedium Bio (VitaDAO portfolio company), using the Prometheus dose-adjustable platform.
October 2025: Eli Lilly participated in NewLimit's additional $45M financing after its Series B, entering partial epigenetic reprogramming.
March 2026: Eli Lilly expanded its Insilico Medicine collaboration to $2.75B ($115M upfront), with exclusive worldwide license to a portfolio of preclinical oral therapeutics.
Any other major deal that should be on this list?
Preprint Corner — in collaboration with The Longevist
The Longevist is a preprint overlay journal spotlighting the most promising longevity research. The journal is currently paused as we build an automated curation agent - the Longevist AI Agent (LAIA). To stay updated, sign up to our newsletter and follow us on X.
Radical No More: Societal Perceptions of Life Extension – Past, Present, and Future Directions
Many cultural stories portray life extension as dangerous or immoral, but this likely reflects old psychological coping mechanisms rather than true opposition to living longer. In reality, most people support extending healthy lifespan, especially if it’s safe and fair, suggesting these outdated narratives may be holding back progress.

Temporal AI model predicts drivers of cell state trajectories across human aging
Scientists developed an AI model called MaxToki that can simulate how human cells change over time—like aging—by learning from massive datasets of gene activity. It can even predict new targets to slow or alter aging, some of which have already been experimentally validated.
A multi-omic atlas in the African turquoise killifish reveals increased glucocorticoid signaling as a hallmark of brain aging
A comprehensive study in the short-lived African turquoise killifish found that increased stress hormone signaling in the brain is a key feature of aging. This suggests that chronic stress pathways may play a central role in driving age-related brain decline.

Published Literature Hot Picks
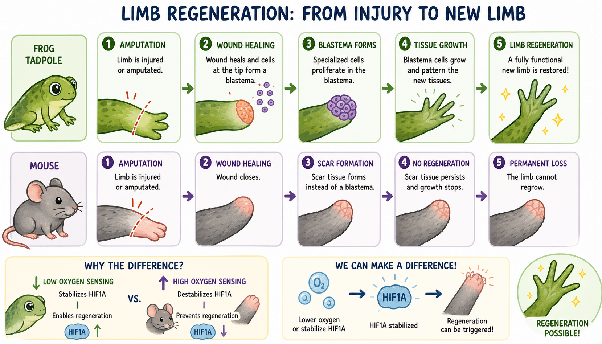
Species-specific oxygen sensing governs the initiation of vertebrate limb regeneration and Hyaluronic acid and tissue mechanics orchestrate mammalian digit tip regeneration
Some animals can regrow limbs because of how their cells sense oxygen and the environment around them: low oxygen signaling and a soft, flexible tissue matrix enable regeneration, while high oxygen sensitivity and stiff, scar-like tissue block it. Encouragingly, tweaking these conditions in mice, by stabilizing key pathways or altering tissue structure, can partially switch on regenerative ability.
Multitissue, multi–time point transcriptomic atlas of aging in mice and rats
Researchers created a large gene expression atlas across the lifespan of mice and rats, revealing that aging affects different tissues, sexes, and species in distinct ways—and at different times. This resource helps pinpoint when and where key aging-related changes occur, offering a roadmap for targeting interventions.
Human immune aging clock identifies RUNX1 as a decelerator of T cell senescence
Scientists built a detailed “immune aging clock” from human blood cells, showing how the immune system declines with age—especially through loss and dysfunction of T cells. They identified a key gene, RUNX1, that helps keep immune cells youthful, and restoring it can reverse signs of aging in these cells.
Redox rhythms promote fitness by modulating ageing-dependent reprogramming
Aging disrupts the body’s daily biological rhythms, especially redox (oxidation) cycles, which contributes to declining health. In mice, restoring these rhythms at the right times improved metabolism, physical function, and even reversed some age-related molecular changes.

Dynamics of genetic and somatic trade-offs in ageing and mortality
A large mouse study shows that genes influencing lifespan don’t act in a fixed way, they can help or harm survival depending on age and sex, with effects often flipping over time. This highlights that aging is shaped by dynamic, stage-specific genetic networks, which could guide more targeted longevity interventions.
Single-cell analysis of the human immune system reveals sex-specific dynamics of immunosenescence
A large human study shows that men and women’s immune systems age differently, with women showing stronger shifts in immune cell activity and men showing changes linked to certain disease risks. These findings highlight the need to tailor healthy aging strategies to biological sex.
Cell-type-specific transposon demethylation and TAD remodeling in aging mouse brain
Scientists created a detailed map of how the brain changes with age at the molecular level, revealing that different cell types and regions age in distinct ways. These shifts in gene regulation and DNA structure could help explain, and eventually predict, neurodegenerative disease risk.
Pleiotropy and disease interactors: the dual nature of genes linking ageing and ageing-related diseases
Genes linked to aging don’t usually cause specific diseases directly, instead, they act more like central regulators that influence many conditions across the body. In contrast, disease-specific genes tend to act locally, affecting particular tissues or immune pathways.
High altitude–mediated immune remodeling accelerates aging
Living at very high altitudes may accelerate aspects of aging, particularly in the immune system, with people showing higher levels of “aged” immune cells and signs of faster biological decline. These effects were also seen in mice, suggesting low-oxygen environments can speed up aging-related changes in the body.

Aging disrupts sympathetic innervation of the thymus
The thymus, which helps produce immune cells, deteriorates with age partly because its nerve supply breaks down. Restoring or mimicking these nerve signals in mice improved thymus function, suggesting a new way to support the aging immune system.
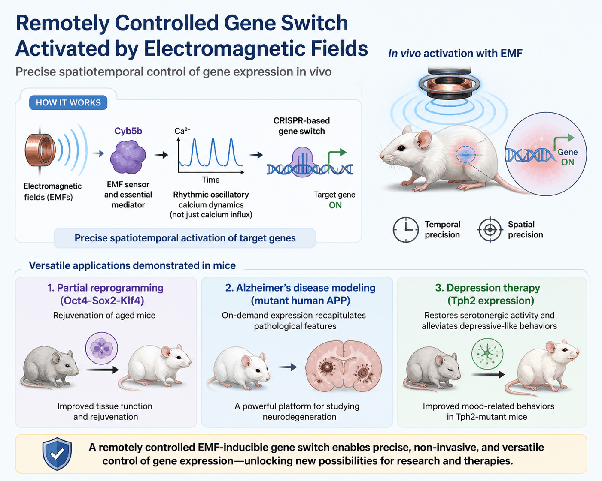
Electromagnetic field-inducible in vivo gene switch for remote spatiotemporal control of gene expression
Scientists have developed a way to switch genes on inside the body using electromagnetic fields, essentially a remote control for gene activity. In mice, this approach could rejuvenate tissues, model Alzheimer’s disease, and improve depression-like symptoms, highlighting its potential for precise, non-invasive therapies.
Published Literature Reviews, Comments, Perspectives and more
The case for space as a model of accelerated aging
Spaceflight exposes astronauts to extreme conditions that trigger many of the same biological changes seen in aging—often much faster. This makes astronauts a powerful real-world model for understanding how environmental stressors drive aging and how we might slow it down.

Senescence in cancer: Hallmarks, paradoxes, and therapeutic promise
Cellular senescence is when damaged cells stop dividing and change how they behave, sometimes helping with healing and cancer prevention, but also contributing to aging and disease. Because its effects vary widely depending on context, scientists are now exploring how to selectively harness or remove these cells for better health outcomes.
Target identification and assessment in the era of AI
AI is speeding up drug discovery by helping scientists identify promising drug targets much faster and more accurately than traditional methods. While challenges remain, these tools are already helping bring new treatments into clinical trials.
Sex differences in response to longevity interventions
Anti-aging interventions don’t affect everyone the same, men and women can respond quite differently due to biological differences like hormones and metabolism. Understanding these differences could help tailor more effective, personalised strategies to extend healthy lifespan.
The long-lived immune system of centenarians
People who live past 100 often stay healthier because their immune systems age more slowly, resembling those of much younger individuals. Understanding how they maintain this resilience could help develop strategies to extend healthy lifespan for everyone.
Prevailing views of cell senescence overlook its biological complexity
Cellular senescence isn’t just a harmful aging process—it’s also a natural, evolutionarily conserved program that plays important roles in development and normal body function. Recognising this dual role is key to designing better therapies that target senescence without disrupting its beneficial effects.
High-throughput screening for ageing and age-related disease drug discovery: Advances and challenges
Scientists are using tiny, short-lived organisms like worms, flies, and fish—combined with automation and AI, to rapidly test potential anti-aging treatments in living systems. This approach is speeding up discovery, though translating these findings into human therapies remains a key challenge.
Outro
We hope you enjoyed this month’s update on the latest happenings at VitaDAO and across the wider community. Stay tuned for more on what we’ve been working on in next month’s edition.
Further Reading
For the longevity aficionados, here are some additional published research papers to explore:
- Plasma Proteomic Profiling of Young and Older Adults Identifies Candidate Biomarkers of Biological Aging at the Intersection of Age and Disease
- Lifespan stops at death, but when does healthspan stop?
- Five-Year Follow-up of a Multicenter Randomized Controlled Trial Comparing an Aragonite-Based Scaffold With Microfracture and Debridement for Chondral and Osteochondral Knee Lesions
- A targeted epigenetic clock for simultaneous assessment of biological aging and cancer-associated methylation drift
- Polyunsaturated lipid senolytics exploit a ferroptotic vulnerability in senescent cells
- Progerin expression in humans: Implications for natural ageing
- p21+TREM2+ senescent macrophages fuel inflammaging and metabolic dysfunction-associated steatotic liver disease
- Engineered immunosuppressive dendritic cells protect against cardiac remodelling
- New approach methodologies for drug discovery
- Acute Finnish sauna heat exposure induces stronger immune cell than cytokine responses
- The longevity effects of reduced IGF-1 signaling depend on the stability of the mitochondrial genome
- Exoproteome of calorie-restricted humans identifies complement deactivation as an immunometabolic checkpoint reducing inflammaging
- Vitamin C inhibits ACSL4 to alleviate ferro-aging in primates
- Double-Pronged NAD Preservation: Delaying Cellular Senescence and Initiating Musculoskeletal Regeneration
- SRN-901, a Novel Longevity Drug, Extends Lifespan and Healthspan by Targeting Multiple Aging Pathways
- Deciphering the role of SIRT6 in suppressing the AMPK-mTOR-TFEB axis: regulation of autophagy activation in HCC




