A single-cell transcriptomics study identifies therapeutic targets for hematopoietic stem cell rejuvenation VitaSTEM — The first AI-enabled longevity project and inaugural IP-Token launched by Aubrai,
A single-cell transcriptomics study identifies therapeutic targets for hematopoietic stem cell rejuvenation
VitaSTEM — The first AI-enabled longevity project and inaugural IP-Token launched by Aubrai, VitaDAO’s decentralized scientific agent.
850K+ cells analyzed 201 human donors 23–91 age range 11 priority targets
Hematopoietic stem cells sustain our blood and immune systems throughout life, yet their regenerative capacity declines with age. This decline—driven by mitochondrial dysfunction, DNA damage accumulation, and inflammatory signaling—underlies immunosenescence and predisposes us to age-related disease. VitaSTEM set out to map the transcriptional changes underlying this decline and identify druggable targets for intervention.
The project integrated three public single-cell RNA sequencing datasets covering both circulating and bone marrow-resident HSC populations across 201 healthy donors aged 23 to 91 years. Using Seurat for clustering and Harmony for batch correction, the analysis identified consistent age-related gene expression patterns across all three datasets—patterns that converge on two core mechanisms.
Two Mechanisms of HSC Aging
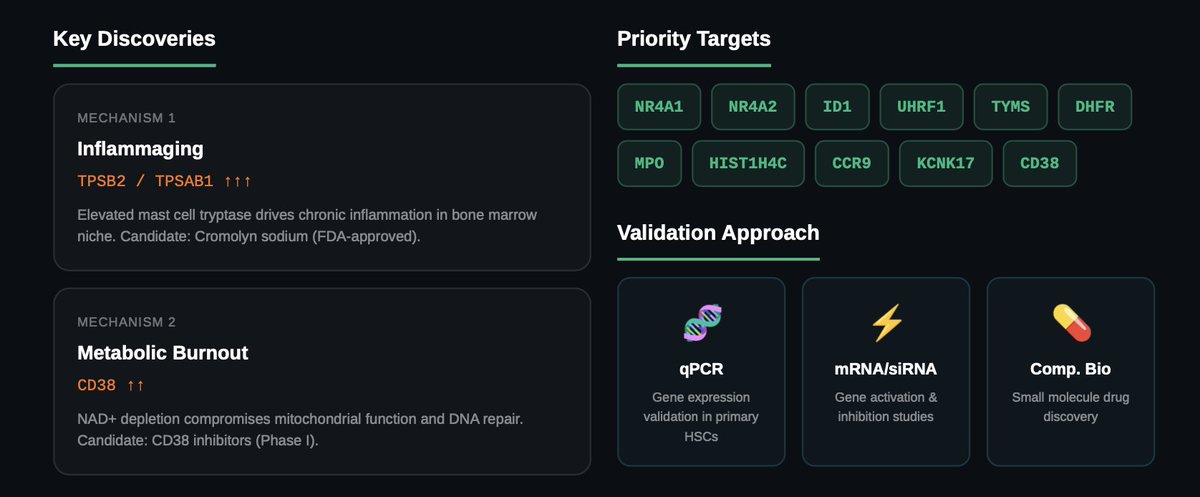
- Inflammaging. The analysis revealed elevated expression of TPSB2 and TPSAB1—mast cell tryptases that drive chronic low-grade inflammation in the bone marrow niche. This inflammatory environment disrupts HSC quiescence and compromises regenerative capacity. Cromolyn sodium, an FDA-approved mast cell stabilizer, emerges as an immediate repurposing candidate.
- Metabolic burnout. Overexpression of CD38, an ectoenzyme that depletes NAD+, was consistently observed in aged HSCs. NAD+ depletion compromises mitochondrial function, DNA repair, and cellular energy metabolism. CD38 inhibitors currently in Phase I development offer a mid-term therapeutic avenue.
Priority Targets
Differential expression analysis identified 11 genes consistently altered between young and aged HSCs. Among the highest-priority targets: NR4A1 and NR4A2, nuclear receptors involved in stress response and metabolism, showed significant downregulation (log₂FC of −4.90 and −3.10 respectively). ID1, a cell cycle inhibitor with established links to HSC exhaustion, showed the largest log₂ fold change at −7.05. Novel targets include UHRF1 (epigenetic regulation), TYMS and DHFR</strong> (nucleotide metabolism), and MPO (oxidative stress). Target prioritization was refined using AUBRAI, a scientific AI agent developed by VitaDAO and BIO Protocol.


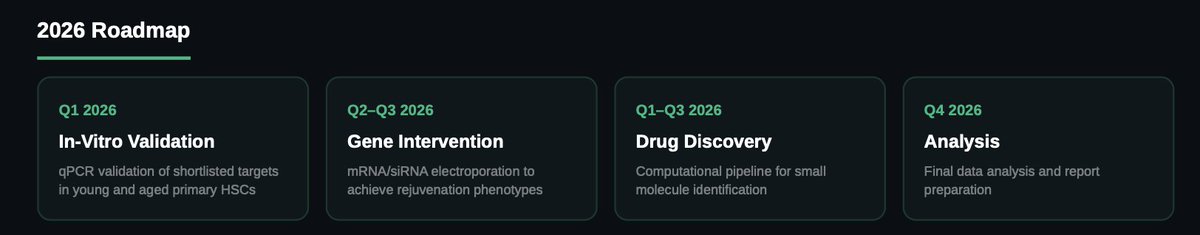
What's Next: VitaSTEM 2026
The next phase focuses on experimental validation. Q1 2026 will validate shortlisted targets via qPCR in primary young and aged HSCs. Q2–Q3 will test gene activation and inhibition using mRNA/siRNA electroporation. In parallel, a computational drug discovery program will identify small molecule alternatives. Final analysis is planned for Q4 2026.
Translational Outlook
VitaSTEM's findings sit within a regenerative medicine market projected to exceed $620 billion by 2030. The project leverages VitaDAO's IP-NFT framework for transparent commercialization. Immediate translational potential lies in repurposing existing drugs—cromolyn sodium for TPSB2 inhibition offers the fastest path to clinical testing given its established safety profile. Mid-term development of CD38 inhibitors and longer-term work on CCR9 agonists and KCNK17 modulators complete a staged therapeutic pipeline.
Principal Investigator: Dr. Ashish Rajput, PhD — Molecular Medicine (German Center for Neurodegenerative Diseases), with postdoctoral training in stem cell biology at TU Munich. 20+ publications, 1,100+ citations. Former CSO at Maximon AG; scientific principal at Longevity Tech Fund.
Source: Embedded X article



